Lithium-ion batteries are a rapid-response solution for energy storage needs. This particular battery technology is a popular with grid-scale applications. Keep reading for an outline how lithium-ion batteries work.
What are lithium-ion batteries?
Lithium-ion batteries are a battery energy storage technology useful for storing and distributing onsite generated power. They can be found in laptops, cell phones and vehicles as well. Lithium-ion batteries are a sought after technology because of their recharging abilities, light weight and high energy density. Energy density refers to the amount of energy a battery can store with respect to its mass and is measured in watt-hours per kilogram (Wh/kg).
Click here for 4 Benefits of Lithium-Ion Batteries
How do lithium-ion batteries work?
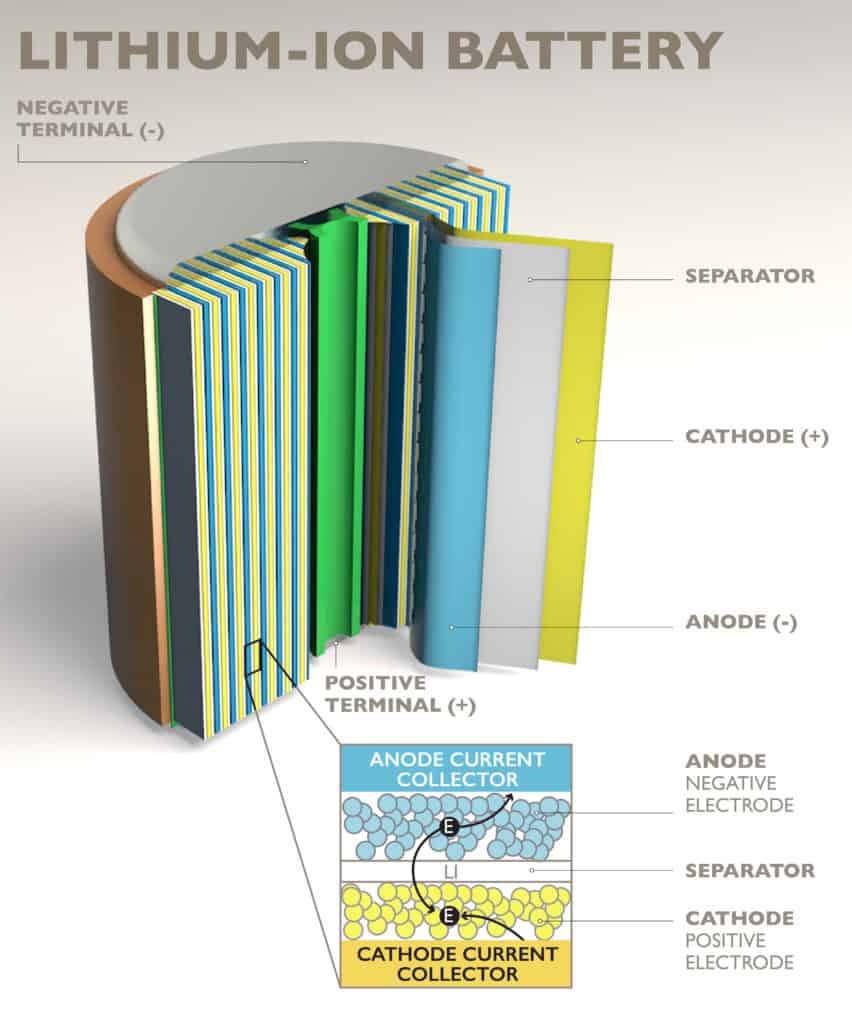
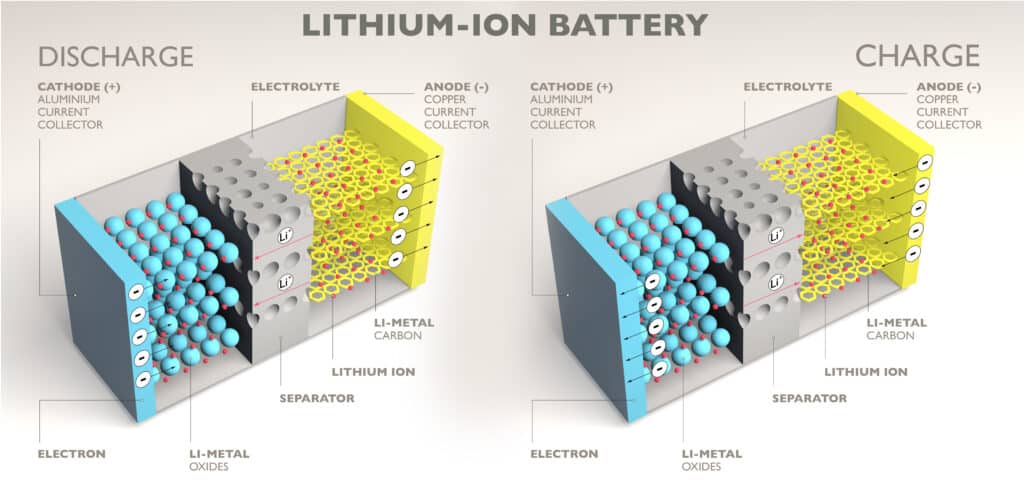
Battery’s are made up of one anode, cathode, separator, electrolyte and positive and negative current collectors. Lithium-ion batteries work by moving positively charged lithium ions from anode to cathode and back through the separator. This flow frees electrons which creates a positive charge and these positively charged ions are carried by an electrolyte. The free electrons cause current to flow through what it is powering and then back to the negative current collector. It is the separators job to block electrons from flowing freely inside the battery. In this particular battery technology, lithium is stored in the anode and cathode.
See the diagrams below for visual representations of the inside of a lithium-ion, one of the battery’s individual parts and one of the battery during both discharge and charge.
Interested in installing lithium-ion batteries?
If you would like to learn more about their installation process now that you’ve learned how lithium-ion batteries work, click below for a free quote. EnergyLink’s team of experts will work to get a rapid-response energy storage solution design, built and funded to fulfill your organization’s unique needs. If you would like to speak directly with a team member, dial (866) 218-0380.






